Stable isotope analysis:
In general, for the general public, the issue of food safety and health has existed in the past and is one of the most important issues in the world today. Since food is the first and most important human priority, therefore, profiteers and fraudsters have a strong tendency to abuse In recent years, unrealistic labels on goods and fraud have endangered not only the rights of consumers but also the health of society.
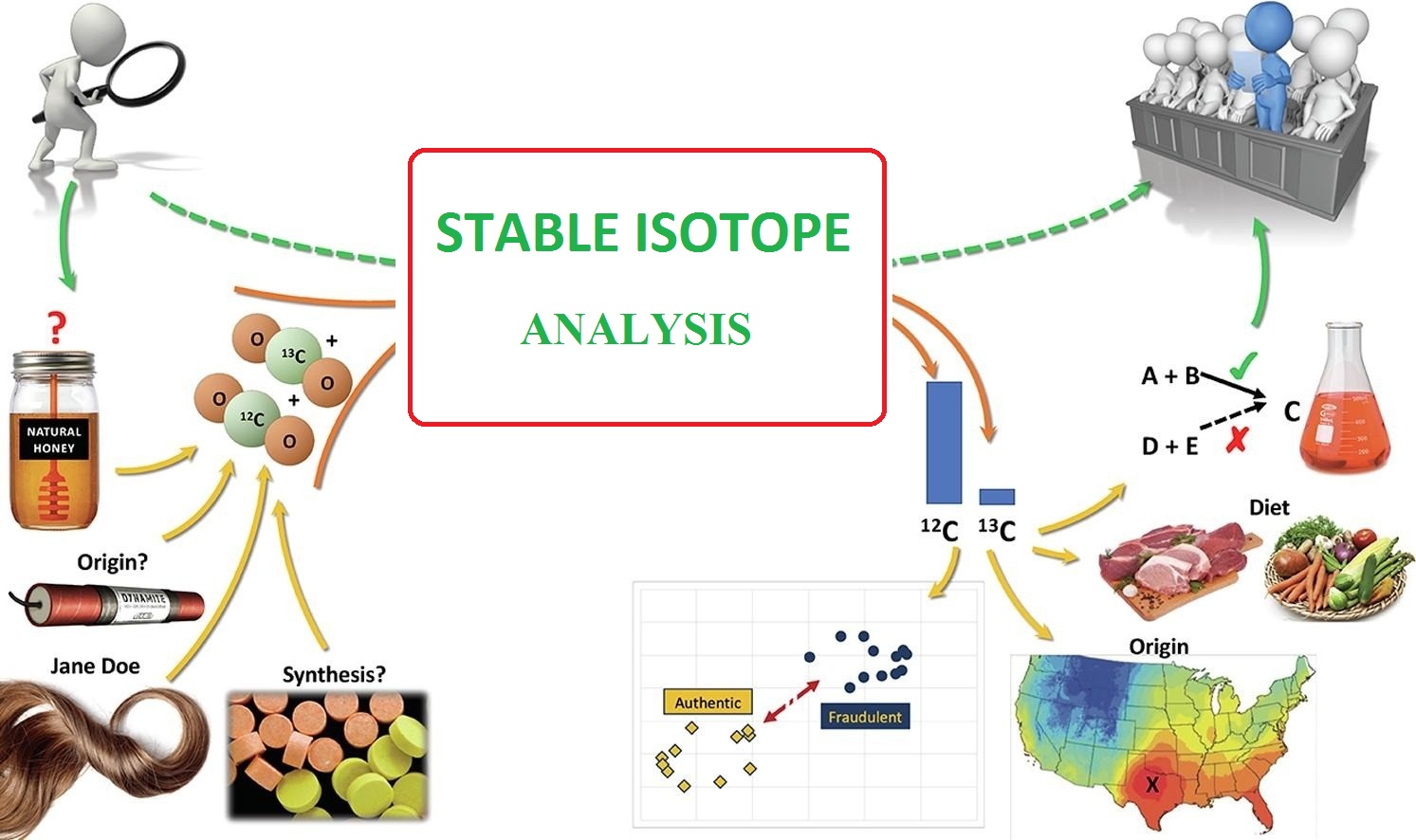
Today, using the IRMS mass spectrometer and examining the isotopic ratio of carbon elements c, nitrogen, oxygen, oxygen, O, etc., the possibility of isotopic fingerprinting and detection of fraud in many foods, including dairy and protein oils, honey, saffron, etc. . has been prepared. The data obtained from the analysis of stable isotopes are like an invisible signature or fingerprint, which can not be forged and copied without sufficient knowledge and advanced decomposition devices.
Different elements have at least two stable isotopes, usually one of which is predominant in terms of frequency. The isotopic ratio is defined as the ratio of the number of heavier to lighter isotope atoms. These ratios are mainly controlled by plant metabolism and animal diet. These ratios can be measured by the Stable Isotope Ratio Mass Spectrometer (IRMS).
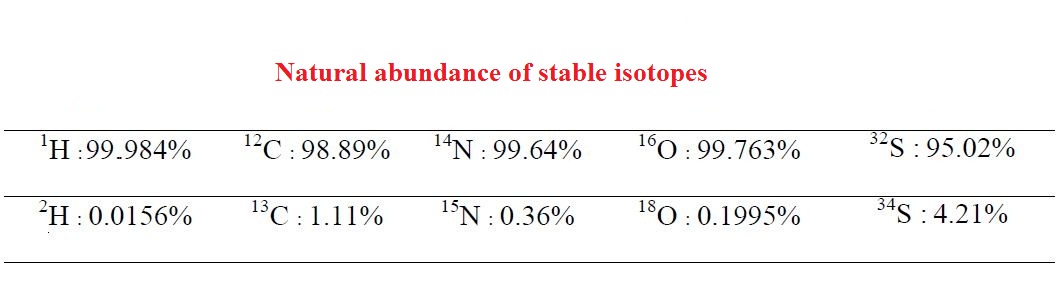
Application of stable isotopes in the study of food originality
In general, the isotopic ratio extracted from the plant reflects the effect of various factors, including the method of cultivation and fertilization of materials absorbed by the plant, reflects the type of culture medium, and the climate of the plant growth environment. For example, different photosynthetic pathways of plants alter the carbon stabilization process, which ultimately changes the isotopic ratio of carbon 13 to carbon 12. Or the isotopic ratio of nitrogen 15 to 14 is largely dependent on soil nutrition and fertilization methods.
The use of isotopic ratio mass spectrometer has been used as an efficient method for detecting food fraud and food criminology. So far, various studies have been conducted around the world, which we will briefly mention:
For example, studies on 40 wheat samples planted in different parts of Canada, China, and Australia showed that by determining the amounts of carbon 13 and nitrogen 15, we can understand the geographical origin of these samples.
Examination of the isotopic composition of carbon 13 in saffron and honey provides a suitable method for determining the origin and geographical origin of these products.
Isotopic analysis is also widely used to detect fraud in livestock and dairy products. For example, isotopic ratio analysis of carbon and nitrogen can reveal the difference between livestock meat produced in different countries and the type of animal feed (organic or inorganic).
Similar studies on the isotopic composition of cheese have shown that the isotopic composition of cheese can be strongly correlated with the amount of corn consumed in the animal diet, so isotopic studies of meat and agricultural products are widely used for food authenticity.
Carbon and oxygen isotopic data were used to differentiate olive oil produced in different regions. Also, by measuring carbon 13 related to fatty acids, the degree of quality and origin of palm oil can be determined.
KEYWORD SECTION:
You can also find other articles about the above keywords in other languages with the use of these words.
Stable isotopic analysis : In Chines: “稳定同位素分析 “ , In Russian: анализ стабильных изотопов