What is Oxygen Isotope - 18?
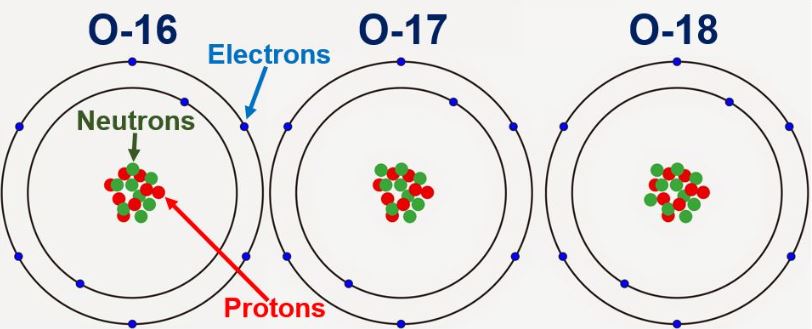
Oxygen is the most abundant element in the Earth's crust, like all nuclear elements of protons and neutrons surrounded by clouds. All oxygen atoms have 8 protons, but the nucleus may contain 8, 9, or 10 neutrons. The lightest oxygen isotope (Oxygen-16) with 8 protons and 8 neutrons is the rarest and most abundant oxygen isotope in nature, followed by much less oxygen - 18 as the heavier isotope, with 8 protons and 10 neutrons. Meanwhile, the stable isotope oxygen-18, despite its small amount in nature, is widely used in various sciences such as the environment; biochemistry; Diagnosis, and medical treatment.
Major applications of oxygen-18 isotope
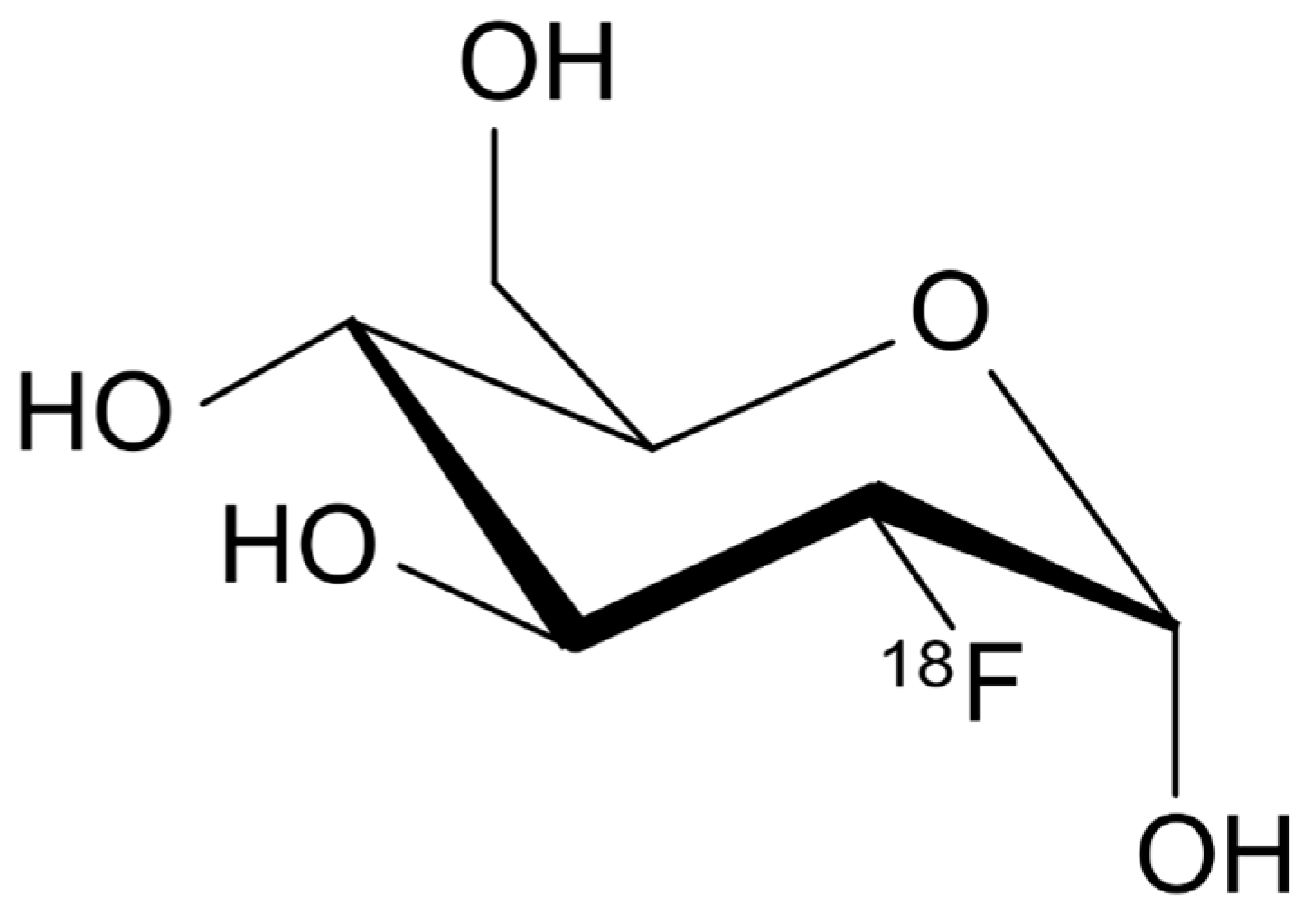
Use of Oxygen-18 Isotope as Precursor for Fluorine-18 Radioisotope Production [18F]
The use of oxygen-18 isotope in medical sciences to diagnose diseases using the technique of detecting pet scan or cut-off with positron emission has led to the development of one of the most accurate methods of scanning cancer cells in the earliest stages of growth. The main application of oxygen-18 is in the form of an H218O molecule. This isotope is used to produce an 18F radio tracker to synthesize an 18FDG labeled sugar molecule in a device called PET scan to detect cancerous tumors and determine the progression of the disease. Diagnosis of brain diseases such as epilepsy and Alzheimer's is used to diagnose heart disease.
Use of oxygen-18 isotope in environmental studies
Due to the development of different methods of isotopic analysis, the possibility of effective use of compounds labeled with stable isotopes has been achieved in many environmental applications. For example, the use of sulfur dioxide, labeled with oxygen-18, makes it possible to identify the source of acid rain.
Use of oxygen-18 isotope in chemical and pharmaceutical research
By marking chemical compounds with oxygen-18 isotope and tracing it in chemical processes and reactions, the mechanism of reaction and study of physical-chemical processes can be studied. O-gas with O2 form is also used in the synthesis of labeled hydrocarbon compounds and as a tracer in studies of materials science and plant biology.
stable oxygen isotope enrichment -18
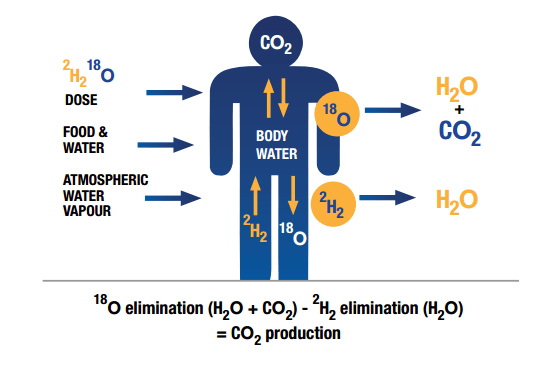
D218O
In fact, oxygen-18 exists in nature and is sufficient to isolate it from other stable oxygen isotopes to produce it. In the process of producing oxygen-18, first, its intermediate product is enriched in the form of a D2 18O molecule. This product is used as doubly labeled water by deuterium and oxygen-18 isotopes to study the metabolic rate of living organisms. In this method, animals are fed with D2 18O for a few days with a certain isotopic richness. Deuterium is excreted from the body by water, urea, and oxygen-18 by water and CO2. By determining the isotopic ratio of these compounds, the body can be metabolically studied and the amount of CO2 produced can be measured.
H218O
in the production of high-consumption isotopic species H218O, first, the D2 18O molecule produced in the distillation process, after a step of the electrolysis process, is decomposed into D2 and 18O2 molecules. Among the applications of D2 gas, it can be used to increase the efficiency of transistors, optical fibers, computer memory, HPLC arc lamps, etc. Then, in the next step, 18O2 molecule is combined with pure H2 molecule in a chemical reaction, and H218O product is produced.
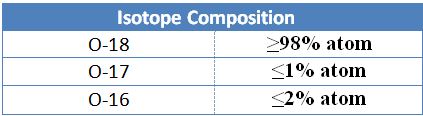
At present, Mesbah Energy Company can produce Oxygen-18 products with isotopic richness above 98%. This product is packaged and distributed in 20 and 50-gram packages in standard borosilicate glass vials with rubber caps and aluminum seals that have been sterilized before filling. You can see the product specifications in the table below.

Click to buy "water-18o", "isotope oxygen 18","H218O" , "D218O"
KEYWORD SECTION:
You can also find other articles about the above keywords in other languages with the use of these words.