Main Menu
Other Sections
About Us
Loading
Blog

Application of NMR in the food industry
NMR is able to analyze the physical and chemical properties of different systems at the molecular level. The basis of NMR work is the analysis of the nuclei of atoms that ...
Read More
Meteorology, oceanography and climate change using stable isotopes
Climate change is one of the greatest threats to human civilization, but determining future climate scenarios is only possible if we know what has happened to the planet's climate in the past. By understanding the changes ...
Read More
Application of stable isotope technique in ecological studies
In recent years, with the development of nuclear science, the application of isotopic techniques (stable and radioactive) in biological and non-biological sciences has grown significantly, while in the field of biological sciences
Read More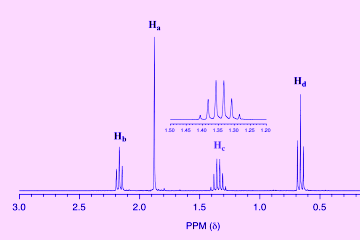
Signal intensity in the 1H-NMR spectrum
Many modifications can be obtained from H-NMR. The first data is the amount of chemical displacement. With the help of this information, we determine the type of hydrogen atom that produces the signal ...
Read More
Application of stable isotopes in determining the authenticity of natural honey using IRMS method
Fraudulent honey production seems to be one of the major problems threatening the survival and activity of the beekeeping industry. Unfortunately, the prevalence of this phenomenon in some countries ....
Read More
Use of Deuterated compounds as an internal standard in newborn screening using LC-MS / MS
Newborn screening is a screening test that is performed for early detection of abnormalities or congenital metabolic diseases, in order to identify the disease at birth ...
Read More
Application of stable isotopes in the measurement of steroid hormones using the LC-MS / MS system
According to the researches and experiments done in the field of material analysis by LC-MS / MS method for quantitative and accurate measurement and high sensitivity of steroids, like fatty acids, it has been used as an internal standard.
Read More
Importance and benefits of nuclear magnetic resonance spectroscopy in H NMR-assisted metabolic research
Metabolomics as an interdisciplinary research field has attracted the attention of many scientists in recent years. One of the most powerful tools in metabolic studies is the magnetic resonance of the NMR nucleus
Read More
Buy NMR Solvents ( Deuterated Solvents for NMR )
Buy common NMR solvents with excellent chemical purity.
Read More